Structural biology, biochemistry, and physiology of neuron/cancer signaling
Overview
Higher-order brain functions, including learning and memory formation, result from cellular signal transduction processes driven by the assembly of macromolecules in neurons that respond to environmental stimuli. Dysfunction within this macromolecular machinery is often linked to neurological disorders such as schizophrenia, epilepsy, stroke, depression, Parkinson’s disease, and Alzheimer’s disease—conditions that continue to present significant and debilitating clinical challenges. Our broad research interests focus on critical cellular paradigms in neuroscience, such as neuroplasticity and neurodegeneration, which are mediated by alterations in membrane potentials and dynamic protein-protein interactions. Recently, we have also expanded our scope to explore the biology at the interface between neurons and cancer in the context of autoimmunity. Our research encompasses (1) the biochemistry and structural biology of membrane-embedded or membrane-associated receptors and their interacting signaling molecules by cryo-EM and x-ray crystallography, (2) functional analyses of ion channels through electrophysiological techniques, (3) protein engineering for the development of reagents for therapy, detection, and diagnosis, (4) experimental and computational approaches for compound development, (5) imaging of protein complexes using both light and electron microscopy, and (6) understanding interactions between cancers and neurons in the context of autoimmune disease.
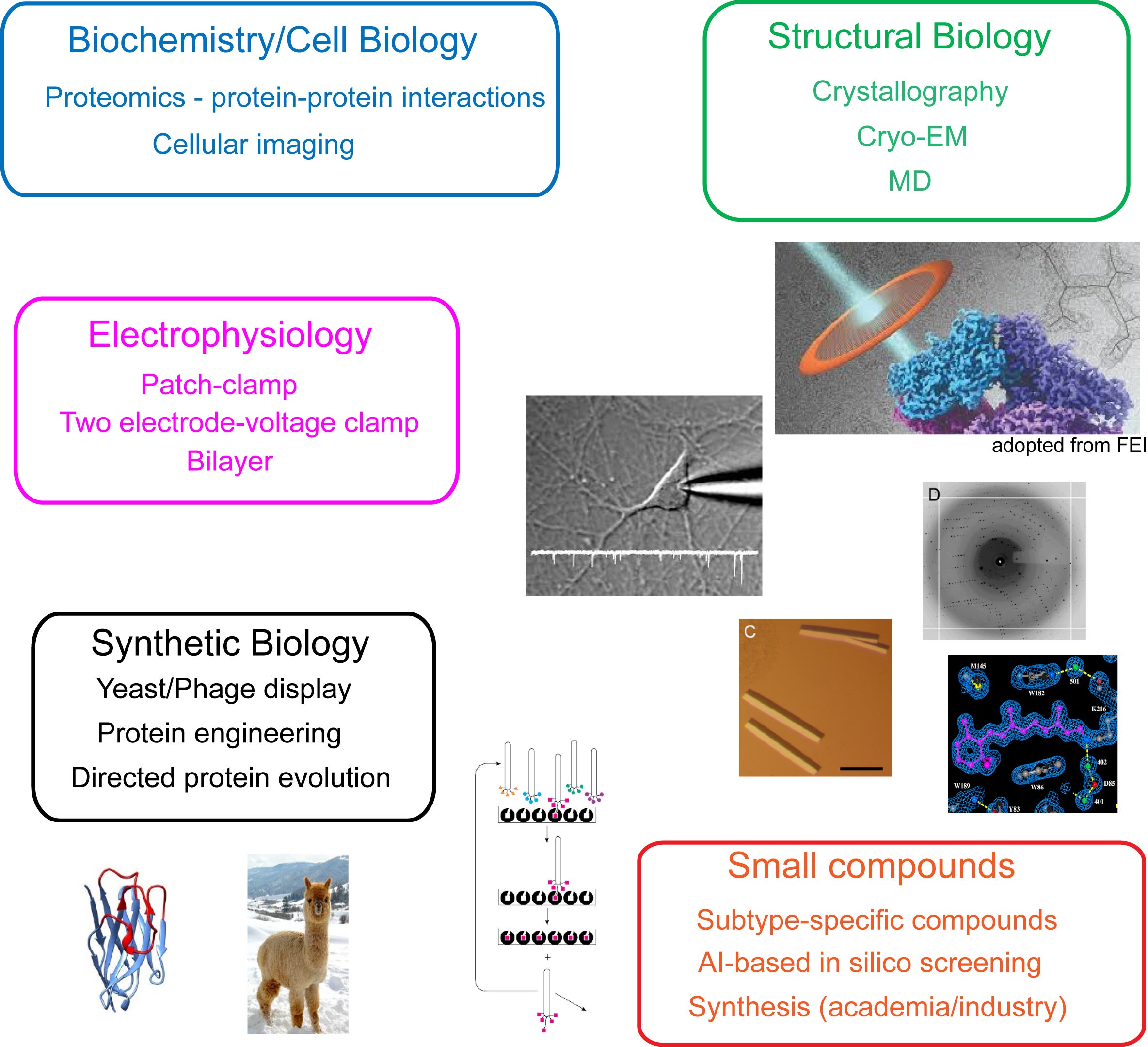
Examples of techniques we implement
Research in the Furukawa lab integrates a multidisciplinary approach to address fundamental biological questions. Our methodologies span structural biology, electrophysiology, biophysics, cell biology, protein engineering, and small molecule development. Key structural techniques include X-ray crystallography and single-particle electron-cryo microscopy (cryo-EM). Additionally, we utilize molecular dynamics (MD) simulations to complement and refine our experimental findings in structural biology and pharmacology. We employ a range of electrophysiological techniques, such as patch-clamp and two-electrode voltage-clamp, to detect and analyze the electrical signals generated by ion channels and transporters. The binding affinities of compound-protein and protein-protein interactions are measured using advanced biophysical techniques, including isothermal titration calorimetry (ITC), and surface plasmon resonance (SPR). We work with the FACS-sorting facility to isolate B-cells and with the Genomic Technology at CSHL for BCR sequencing, spatial transcriptomics, etc. We also work with CSHL’s proteomics facility to identify novel protein-protein interactions within cells. Our synthetic biology efforts, including yeast and phage display and directed protein evolution, facilitate the engineering and production of proteins such as antibodies and proteases. We also collaborate with chemists at CSHL to facilitate our chemical-biological efforts. To support our research, we frequently develop and optimize new methodologies; for instance, we devise the EarlyBac system, which uses the optimal combination of UTRs and promoters to efficiently express and assemble multimeric membrane protein complexes. Hypotheses generated from any of these disciplines are cross-validated using complementary techniques. For example, structural insights can be tested through electrophysiology and biophysical analyses, and findings from these approaches can inform further structural investigations. Below, we provide an overview of some of the current projects underway in our lab.
Structure and Function of intact NMDA receptor ion channels
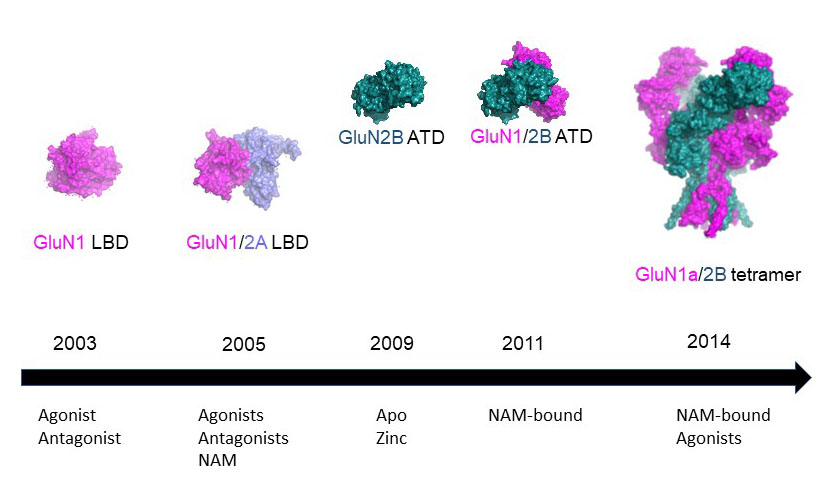
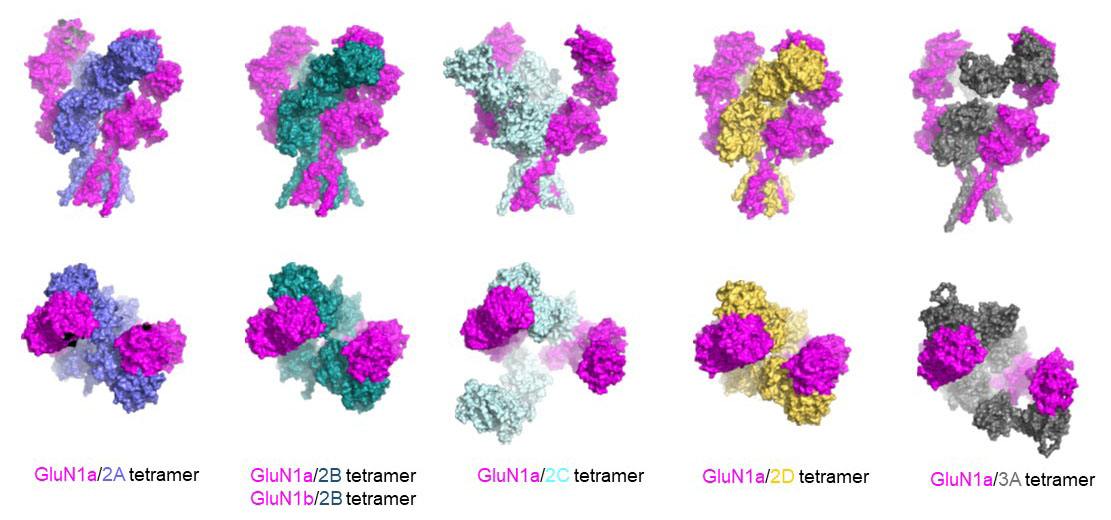
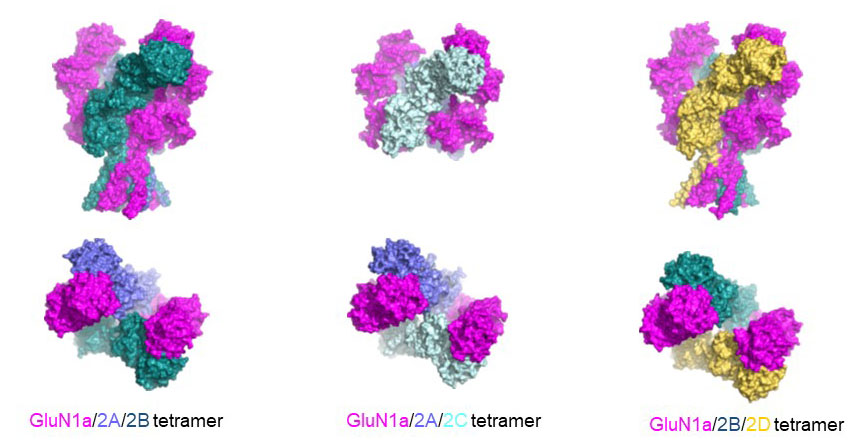
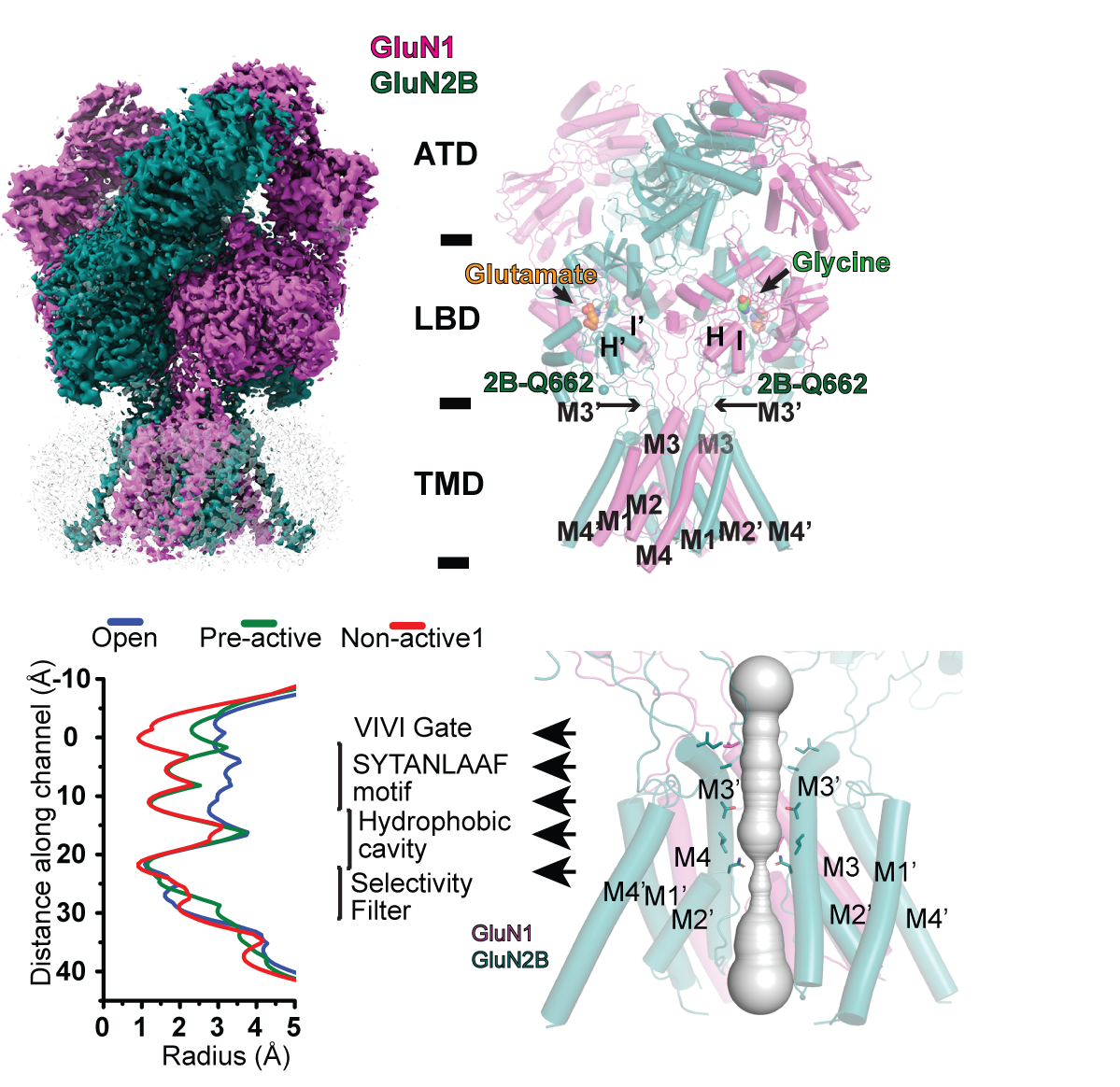
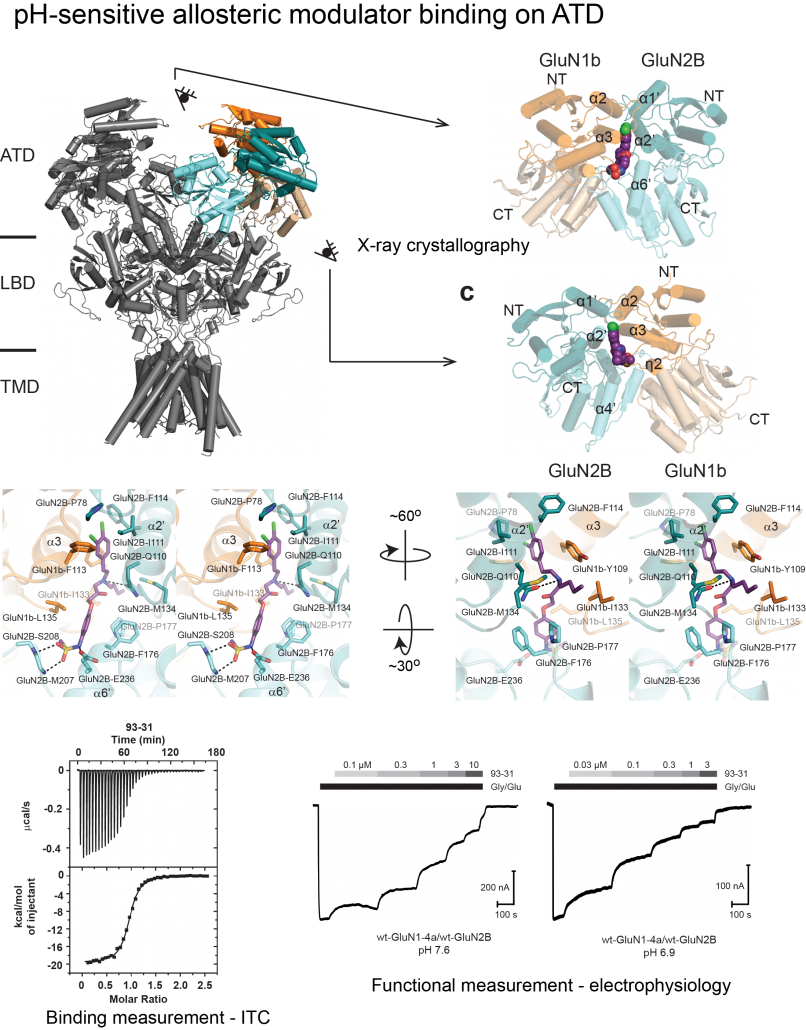
NMDA receptors are heterotetrameric cationic ion channels that are composed of two GluN1 subunits and two GluN2 (A-D) and/or GluN3 (A-B) subunits. GluN1 and GluN3 subunits bind glycine, whereas GluN2 subunits bind the neurotransmitter glutamate. Concurrent binding of glycine and glutamate is necessary for the opening of the cationic ion channel, which contributes to the generation of excitatory postsynaptic potential (EPSP). Furthermore, voltage-dependent calcium influx by NMDA receptors drives cellular signaling for neuroplasticity, which is critical for learning and memory formation. Dysfunctional NMDA receptors are implicated in Alzheimer’s disease, Parkinson’s disease, depression, schizophrenia, stroke, and autism. Eric Gouaux and I initiated structural biology studies of the NMDAR GluN1 ligand-binding domain (LBD) and the GluN1-2A LBD heterodimer (2001-2005). Later, the Furukawa lab captured the GluN2D LBD, the GluN2B amino-terminal domain (ATD), and the GluN1-2B ATD heterodimer by X-ray crystallography. These fragmented approaches were useful in understanding compound bindings and local conformational changes. However, they did not explain inter-domain or inter-subunit conformational movements that regulate ion channel activities in the context of tetrameric channels. In 2014, we solved the crystal structure of the intact heterotetrameric GluN1-GluN2B NMDA receptor from rat in complex with glycine, glutamate, and the allosteric inhibitor ifenprodil. This structure revealed how the extracellular domains, ATDs, and LBDs, from both GluN1 and GluN2 subunits, are associated with each other in the ‘dimer of GluN1-GluN2 heterodimers’ subunit arrangement. Using a combination of X-ray crystallography and single-particle electron cryo-microscopy (cryo-EM), we continue to understand the regulatory mechanisms of NMDA receptors. Specifically, we are working to understand how multiple domains (ATD, LBD, and TMD) move relative to one another across various functional states. The state-of-the-art cryo-EM facility in our building allows us to explore the above at high quality. Indeed, we have resolved the structures of all other subtypes (GluN1a-2A, GluN1b-2B, GluN1a-2C, GluN1a-2D, GluN1a-3A, GluN1a-2A-2C, GluN1a-2B-2D, etc.) using cryo-EM. In combination with electrophysiology, we have been addressing functional questions, including allosteric modulation, pH sensitivity mediated by alternative splicing, competitive inhibition, ligand-gated channel opening, and channel blockade.
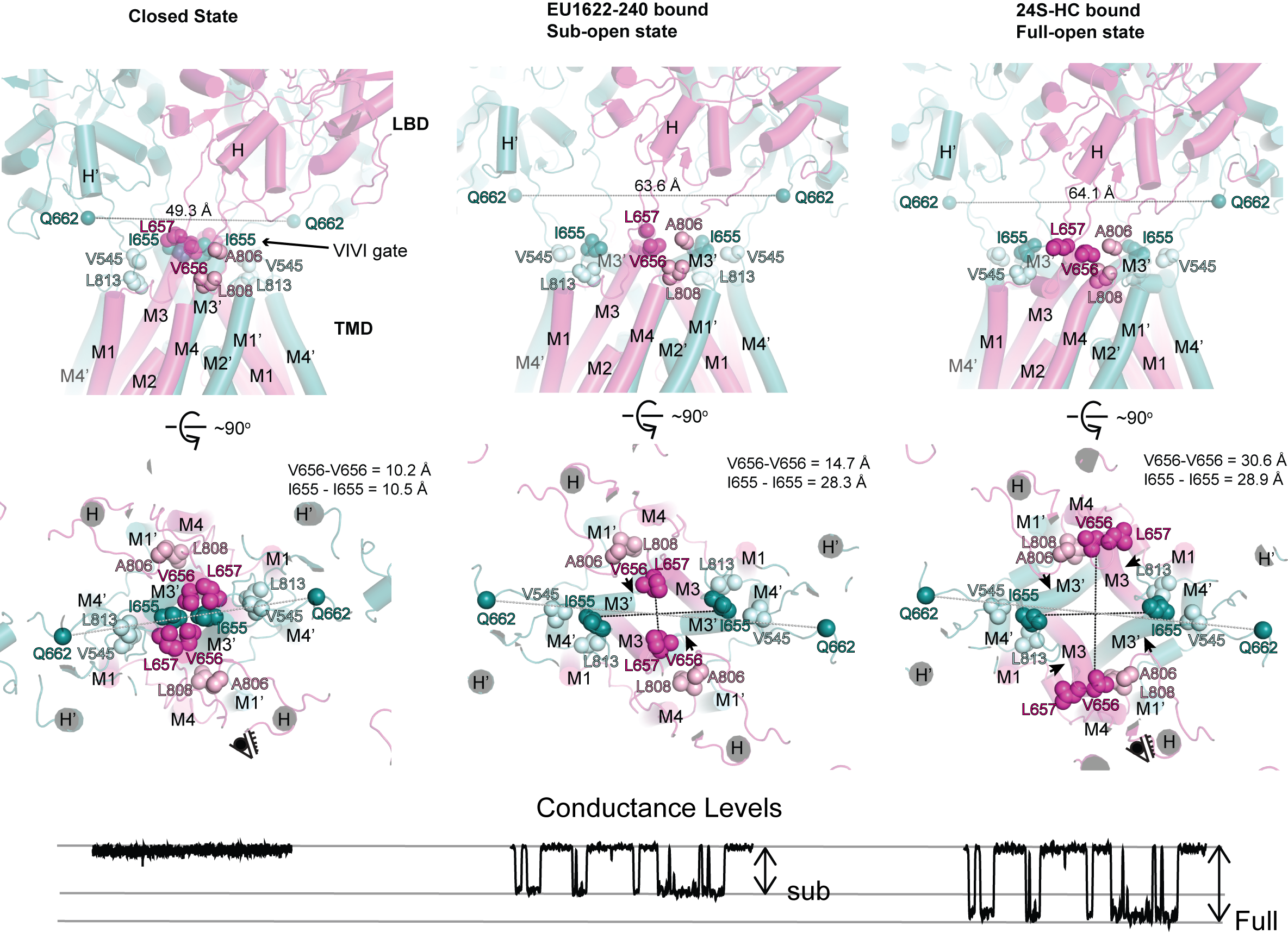
Conformations and Function of intact NMDA receptor ion channels
Over the last few decades, we have monitored conformational changes in both isolated domains and intact NMDAR channels. Therefore, we have some, but not a complete, understanding of the mechanisms underlying activation, inhibition, channel blockade, ligand gating, channel opening, and subconductance levels for some subtypes, but not all. Using a combination of cryo-EM and electrophysiology, we continue to elucidate subtype-specific channel-gating mechanisms.
Structure Pharmacology of NMDA receptors and off-targets
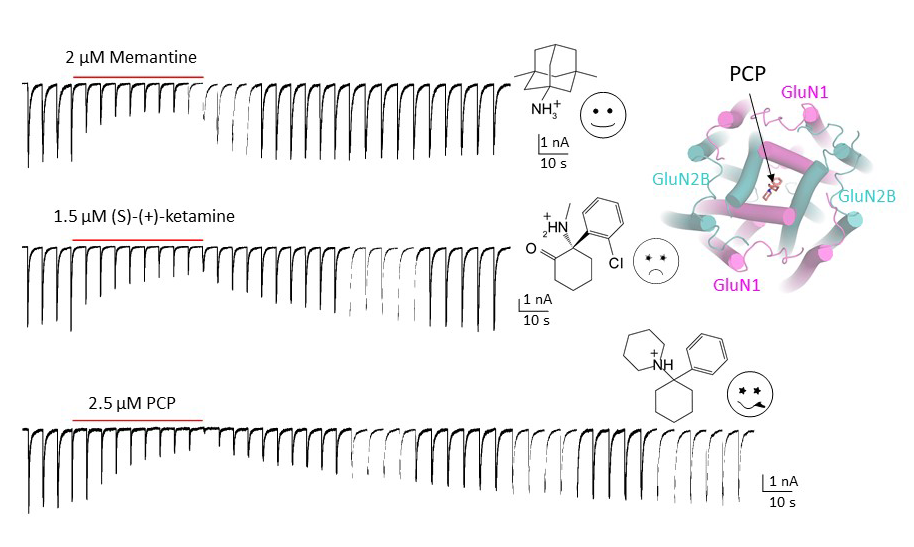
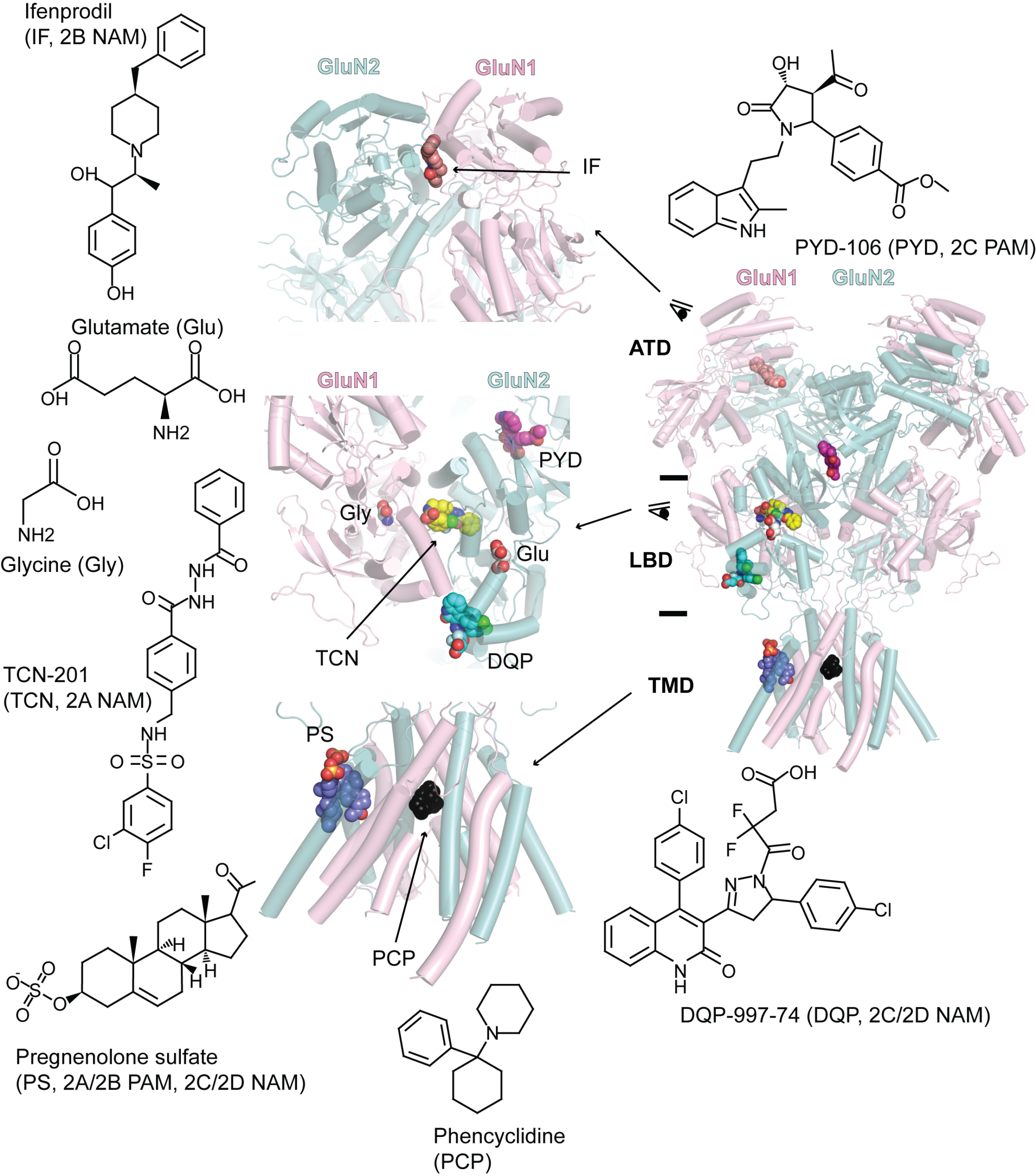
Since the discovery of NMDA receptors in the early 1970s, considerable efforts have focused on characterizing NMDA receptor pharmacology. NMDA receptors exhibit significant diversity: eight splicing variants of the GluN1 subunit can assemble as heterotetramers with four types of GluN2 (A-D) and two types of GluN3 (A-B), forming ion channels with distinct functional properties, including varied open probabilities, opening/closing kinetics, and binding affinities and potencies for compounds. Subtype-specific compounds targeting NMDA receptors hold promise for treating neurological diseases and disorders, making a high-resolution understanding of the molecular determinants of subtype specificity critical. To this end, we use cryo-EM to determine the structures of intact heterotetrameric NMDA receptors, particularly for studying compounds such as channel blockers (e.g., ketamine, PCP, memantine) that interact with the transmembrane domain (TMD). We employ X-ray crystallography for compounds that bind exclusively to the extracellular domains (ATD or LBD). By resolving these domains in complex with target compounds at high resolution (2.0 Å and beyond), we can accurately visualize drug-binding sites, critical water molecules, and conformational changes within these domains. Our objective is to provide structural insights that enable precise mapping of drug-binding sites at atomic resolution, advancing the design of targeted therapeutic compounds in pharmacology. We also aim to identify previously uncharted compound binding sites to uncover novel mechanisms of channel regulation. We also came to realize that some of these compounds target different receptors; therefore, we are characterizing these off-target effects.
Deciphering anti-NMDA receptor encephalitis
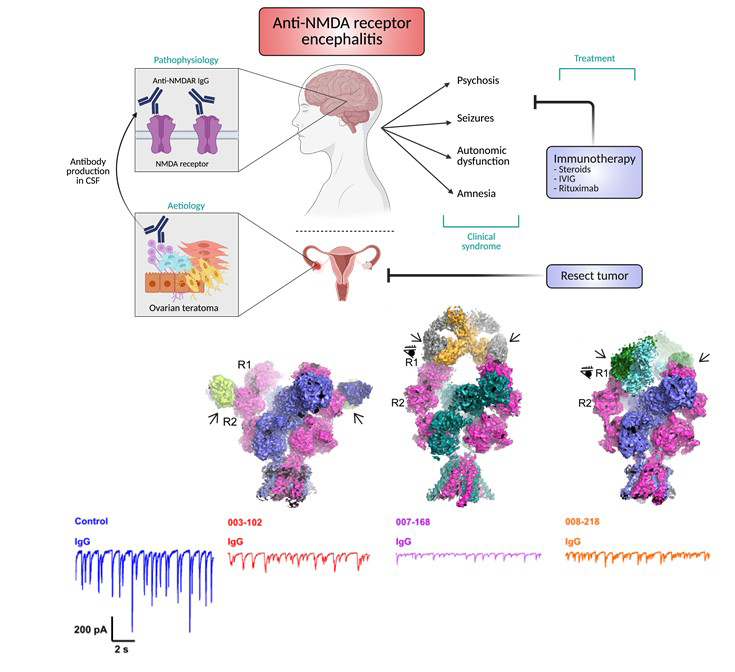
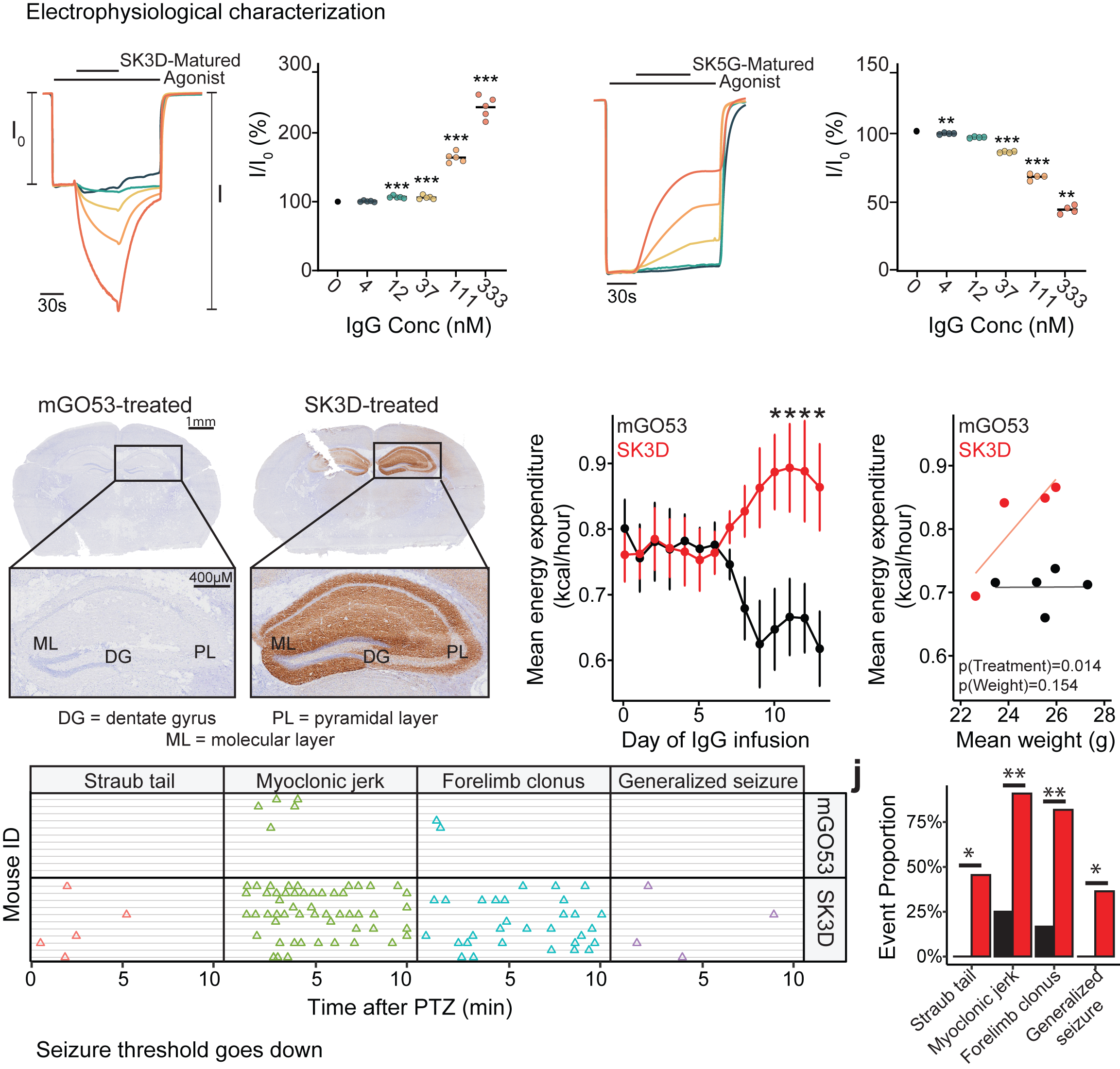
Anti-NMDAR encephalitis is the most prevalent form of immune-mediated encephalitis, predominantly affecting young individuals, typically in their second and third decades of life. Although the NMDAR has been clearly identified as the autoimmune target, the disease’s precise mechanisms remain incompletely understood. This knowledge gap poses significant challenges and is further exacerbated by suboptimal diagnostic methods. Early diagnosis and targeted treatment are essential, as delays in intervention can lead to worsening clinical outcomes, including psychiatric disturbances such as psychosis and behavioral changes, as well as seizures and central hypoventilation. Anti-NMDAR encephalitis is often linked to ovarian teratomas and other cancers that express NMDARs. We examined three IgGs isolated from three patients and demonstrated that they bind to distinct epitopes within NMDAR tetramers. Additionally, we found that these antibodies exhibit varied stoichiometry when binding to the NMDAR tetramers. Our electrophysiological studies revealed inhibition of spontaneous excitatory postsynaptic currents (EPSCs) in cultured neurons. Our findings underscore the epitope diversity and structural and functional heterogeneity of anti-NMDAR antibodies among patients. The Furukawa lab now possesses a robust and effective workflow for testing antibodies isolated from human patients or animal models. This positions us uniquely to elucidate the molecular mechanisms underpinning anti-NMDAR encephalitis and potentially inform new therapeutic strategies.
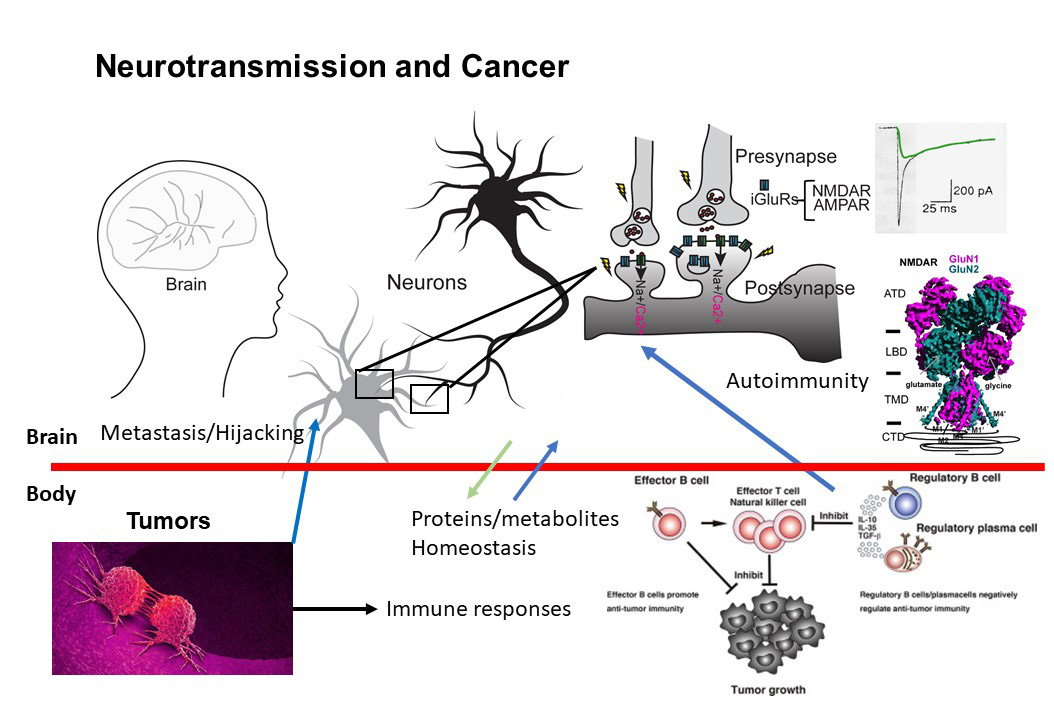
Anti-NMDAR autoimmunity and anti-cancer immunity
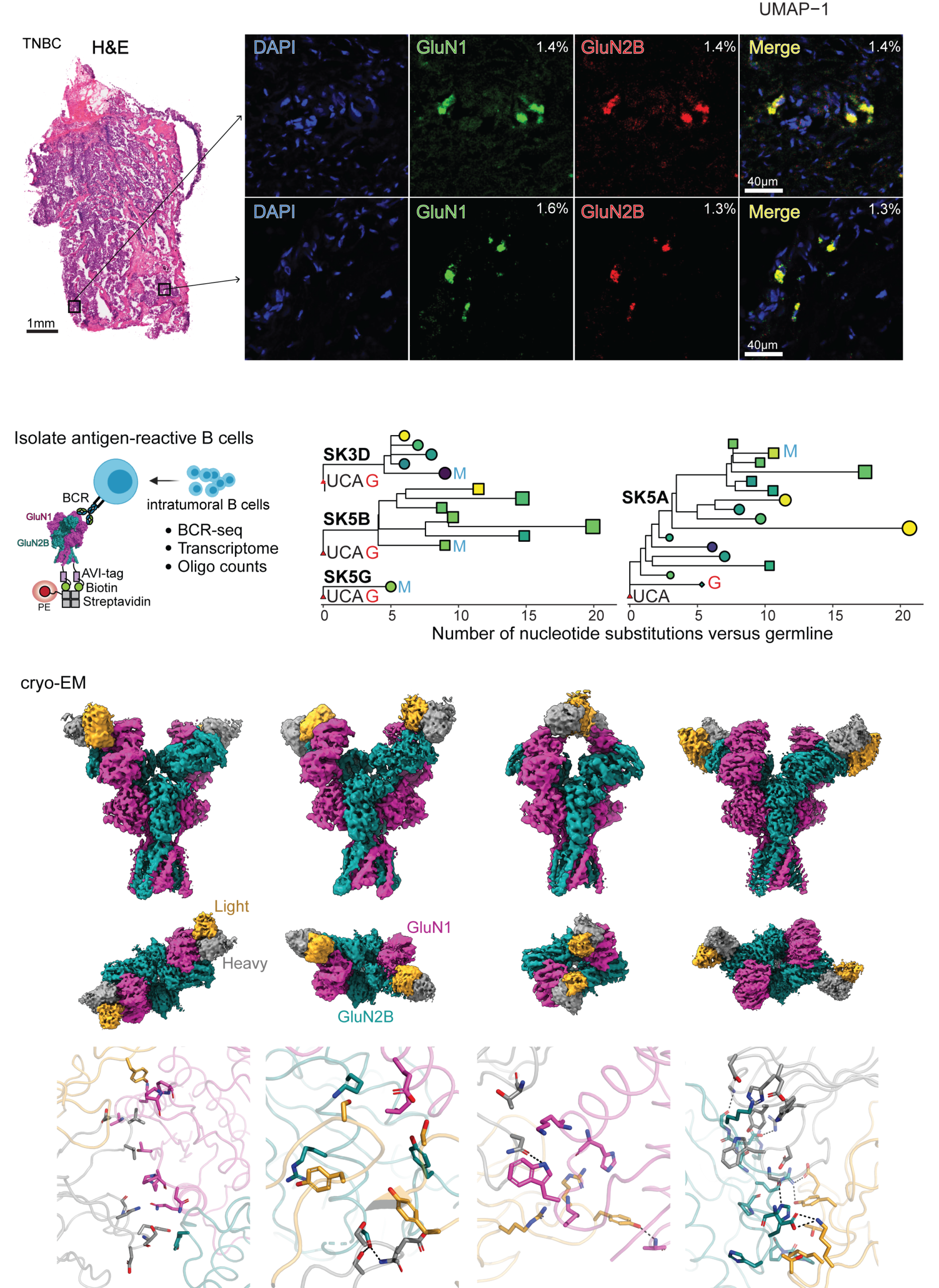
In collaboration with the Janowitz lab at CSHL, we aim to answer a fundamental question about cross-talk between anti-cancer immunity and autoimmunity. We view anti-NMDAR autoimmunity as a paraneoplastic syndrome, in which anti-tumor immunity against NMDAR-expressing tumors is the source of the autoimmune antibodies. NMDARs are normally expressed exclusively in the brain. However, they have been known to express in tumors, including ovarian teratoma, triple-negative breast cancer (TNBC), pancreatic cancer, and lung cancer. We recently engineered a TNBC mouse model expressing NMDAR and showed that anti-NMDAR autoimmunity arises, and these anti-NMDAR IgGs have anti-tumor effects. B-cells expressing anti-NMDAR BCRs are isolated, and the BCR encoding sequences are identified. The epitopes of these antibodies vary as assessed by cryo-EM. The antibodies’ functional effects can be excitatory, neutral, or inhibitory at the level of electrophysiology. The injection of some of these antibodies into the mouse brain manifests in a lowering of the seizure threshold. Therefore, we established an animal model to demonstrate that anti-NMDAR autoimmunity results from anti-tumor immunity. Importantly, the workflow established here is applicable to patients’ samples for diagnosis and potential therapy. We are also exploring autoimmunity against other cell-surface proteins involved in brain functions.
Large-pore channels
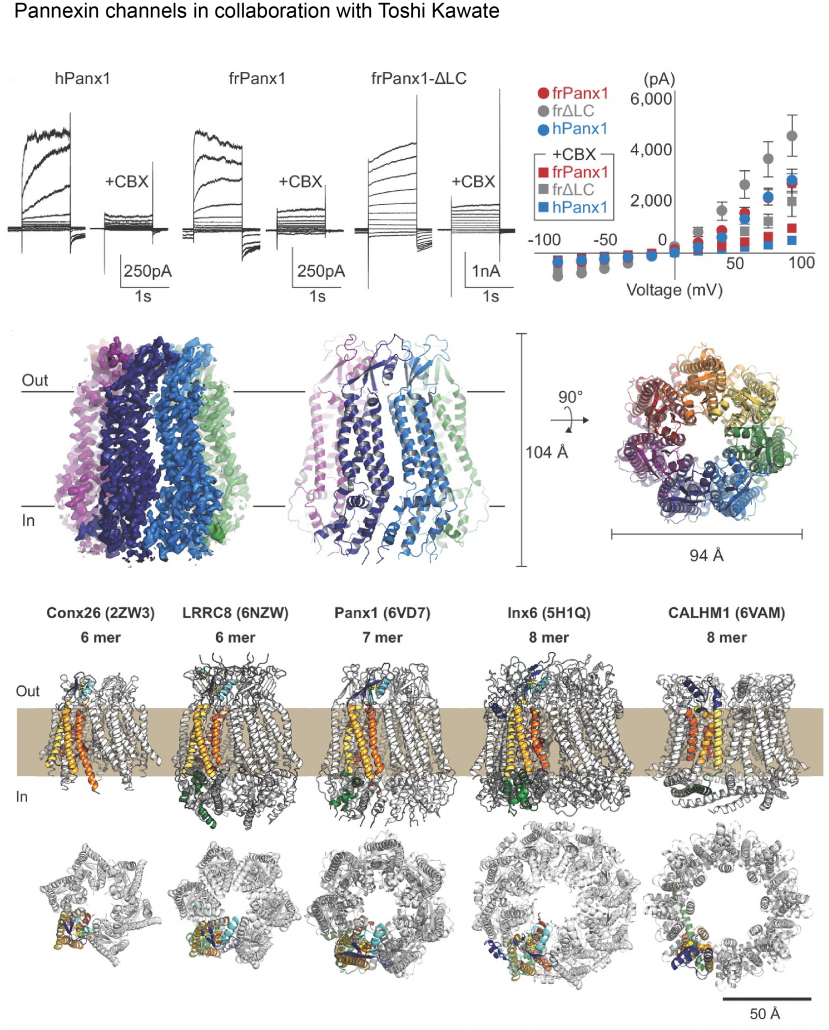
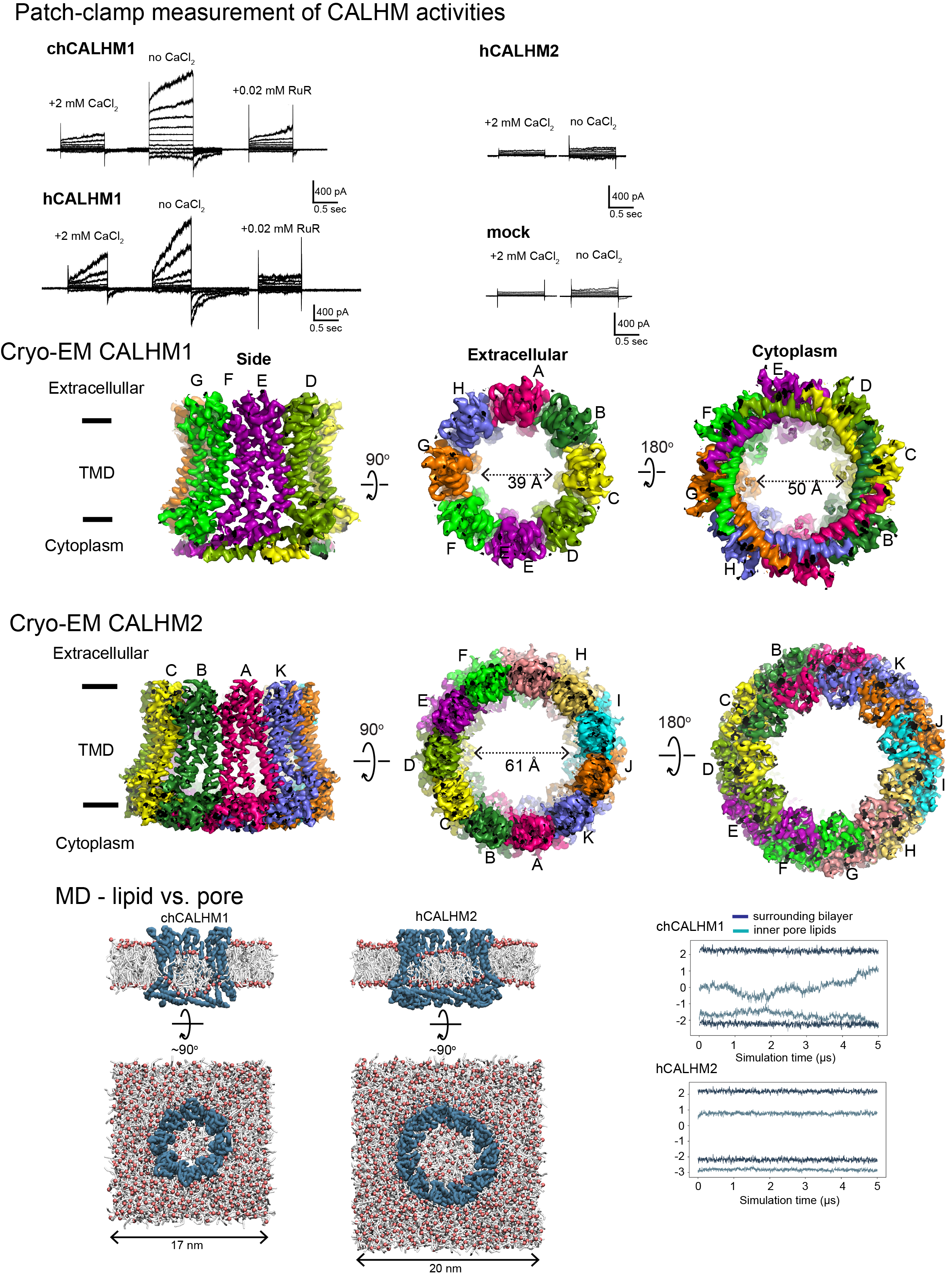
Biological membranes in various tissues and organs contain large-pore channels that facilitate the permeation of diverse ions and metabolites, such as ATP. They are tetra-spanning membrane proteins that form channels of various sizes by forming different oligomers. A classic example is the connexin family and its invertebrate ortholog, innexin, which form gap junctions as hexameric dimers. Other large-pore channels include pannexin, LRRC8, and calcium homeostasis modulators (CALHMs). These channels are mainly expressed on the cell surface as hemichannels (no gap junction formation); therefore, tight regulatory mechanisms are needed to control their activity. These channels employ multiple regulatory mechanisms, such as voltage gating, to maintain cellular homeostasis and viability, thereby supporting various biological functions. Among the most recently discovered large-pore channels are CALHM channels, which enable the voltage-dependent passage of small ions and ATP and play key roles in physiological processes such as taste perception, airway protective chemoreflexes, and cognitive control. SNP in calhm1 has also been implicated in Alzheimer’s disease. We use cryo-EM and electrophysiology to study how CALHM proteins assemble and facilitate the permeation of ions and metabolites across membranes. Additionally, we investigate the structure and function of another large-pore channel, Pannexin, known for its role in ATP efflux.
Structural pharmacology of cancer-related membrane proteins
The cancer biology community at CSHL is known for its highly interactive and collaborative nature. Leveraging our expertise, we are beginning to investigate membrane proteins that play critical roles in cancer cell survival—proteins that can serve as significant vulnerabilities when targeted effectively. Our aim is to elucidate their biochemical properties, structural characteristics, and strategies for therapeutic targeting. Through close collaboration with the Cancer Center at CSHL, the Furukawa lab is set to deepen its engagement in cancer biology.
Reagent development – chemical biological, synthetic biological, and antibody engineering approach
Antibodies are extremely useful biological reagents that can recognize and functionally regulate target proteins. We are screening and developing antibodies for cell surface receptors expressed in neurons and cancer cells. This is done by basic immunization technology and/or the phage- and yeast-display methods. Identified antibodies are further tested for functional effects and are re-engineered using structural information.We further combine this synthetic biological approach with chemical biology in collaboration with John Moses’s lab at CSHL. We are developing reagents that can be used as therapeutic reagents, biomarkers, and tools for basic researches.